This is a guest blog from Sarah White PhD, director of Quit Victoria. Reproduced from Australian Doctor, with permission

The RACGP’s Smoking Cessation Guidelines are generally excellent.
However, I worry the e-cigarette recommendation is not pragmatic and that deliberate misrepresentation of the nuance in the recommendation risks will create a situation in which doctors are faced with an ethical dilemma.
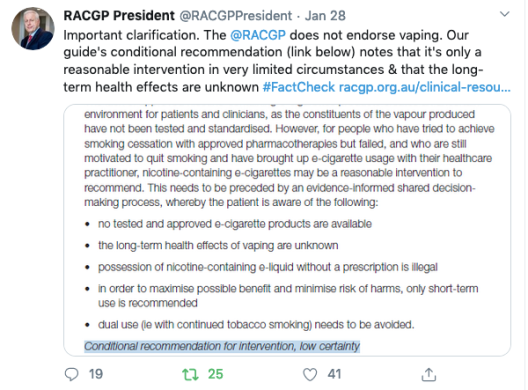
The guidelines acknowledge the lack of approved nicotine-containing e-cigarette products and argue this is creating an “uncertain environment for patients and clinicians, as the constituents of the vapour produced by vaping have not been tested and standardized”.
They add: “However, for people who have tried to achieve smoking cessation with approved pharmacotherapies but failed, and who are still motivated to quit smoking and have brought up e-cigarette usage with their healthcare practitioner, nicotine-containing e-cigarettes may be a reasonable intervention to recommend.”
So is this the college endorsing vaping as a possible second-line therapy for doctors dealing with patients wanting to quit?
To me, the recommendations, when read in context, suggest that doctors should gently dissuade patients from using e-cigarettes through a “shared decision-making process”
It’s important to acknowledge the very real qualifications the guidelines make.
Firstly, they suggest doctors discuss the option only if brought up by the patient themselves when all else has failed.
They also say that doctors should make sure the patient is aware of the following:
- there are no tested and approved e-cigarette products available;
- the long-term health effects of vaping are unknown;
- possession of nicotine-containing e-liquid without a prescription is illegal;
- in order to maximise possible benefit and minimise risk of harms, only short-term use is recommended; and
- dual use needs to be avoided (for example, with continued tobacco smoking).
This advice is not surprising given the current low certainty of evidence. However, more importantly, no products have been assessed as meeting basic Australian consumer safety standards, let alone having gained TGA approval.
There are literally tens of thousands of e-cigarette products available to any patient in Australia, presented by the possible combinations of multiple devices (some with adjustable temperature and electrical resistance settings) with e-liquids that can contain one (or more) of 2000+ chemical flavours, different ratios of carrier and flavouring liquids and a range of nicotine concentrations.
We don’t know which devices shed heavy metals and other chemicals from their interior or which e-liquids contain impurities or contaminants.
We don’t know the by-products created by admixture, pyrolysis and decomposition of e-liquids. Australia’s chemicals watchdog, the National Industrial Chemicals Notification and Assessment Scheme (NICNAS), set out these concerns in a comprehensive—and alarming—2019 review of a small number of non-nicotine e-liquids available for retail sale in Australia.
In my view, the e-cigarette recommendation should have been much less equivocal and based on a synthesis of evidence as it applies to the current retail and regulatory contexts in Australia.
Had this been the case, the recommendation would have been along the lines: “The RACGP is unable to recommend the use of e-cigarettes until a product has been approved by the TGA. If a patient chooses to use an e-cigarette, they should be made aware of the following issues to maximise potential benefit and to minimise potential harm….”
Naturally, inevitably, lobbyists and commercial interests (including tobacco companies) leaped to publicise the RACGP’s new guidelines, conveniently omitting the e-cigarette recommendation’s nuance.
One media release appeared an impressive 35 minutes after the guidelines were issued, claiming the RACGP “endorses” e-cigarettes “in a major policy shift”.
Some hours later, RACGP president Dr Harry Nespolon was forced to tweet an unequivocal “the RACGP does not endorse vaping”.
I note these public relations efforts because I fear that doctors will start seeing patients wanting to discuss e-cigarettes because they have been told by those with vested interests that “doctors recommend them now”.
So, what is a doctor to do if a patient is adamant about trying an e-cigarette in the absence of any safety or efficacy data for a retail product?
Which device type or e-liquid(s) should be recommended? What dose of nicotine should be used? Is it a doctor’s responsibility to inform the patient how to dilute a potent neurotoxin to the right dose?
And how often should the device be used and for what duration? What is the legality of possessing liquid nicotine? (As of December 2019, nicotine possession, even with a doctor’s prescription, is illegal in the Northern Territory.)
Are doctors really going to recommend their patients pop down to the nearest high street ‘Wicked Groovy Vapes’ shop—as advocated by e-cigarette lobbyists—for the answers to these and other questions?
It seems inevitable that doctors will be forced into a choice of either refusing to provide a prescription for liquid nicotine or providing one in the full knowledge their patient could be harmed by how they use it.
To my mind, this presents an ethical dilemma that could have been avoided by a more definitive recommendation along the lines of that issued by Dr Nespolon on Twitter.


Very important that any policy recommendations by leading medical authorities are continually nuanced to provide the most ethical public health response. At the same time its important to acknowledge the rights of patients to seek alternative approaches while doctors should highlight the current lack of evidence on efficacy or real harms of ecigs.
LikeLike